top of page


capsid-based antigen display
Engineering next-generation vaccines through
AdaptVac is a clinical-stage biotechnology company developing a new class of vaccines that deliver broad, durable and adaptable protection.
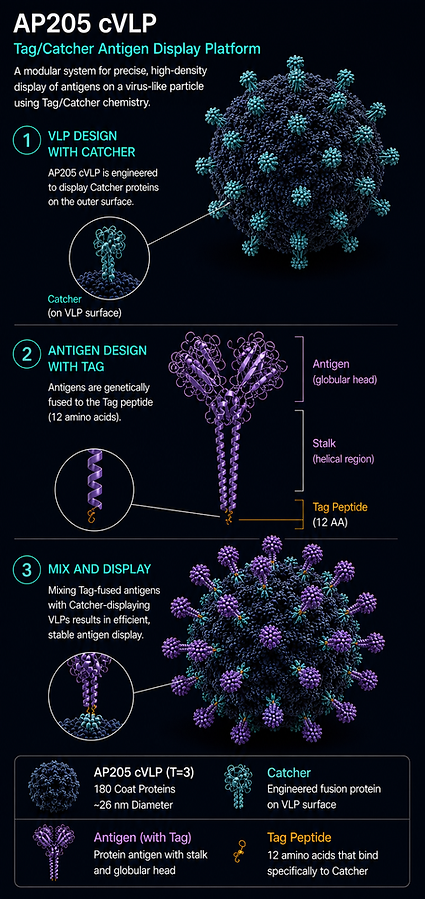
Phase 3 validated Tag/Catcher cVLP display platform
AdaptVac's iso-peptide display platform utilises our proprietary MoonCatcher/Tag pair.
Furthermore, the cVLP is based on Tag or Catcher fused Bacteriophage AP205 capsid protein, which spontaneously forms a capsid Virus-Like Particle during production in E.coli.
MoonCatcher and AP205 have now been through Phase 3 clinical testing in our ABNCoV2 COVID-19 vaccine program.
Demonstrating safety, scalability, as well as EU and US regulatory acceptance for our platform.

bottom of page
